In the popular 2003 Pixar film, Finding Nemo, a clownfish father’s only son is captured by human aquarium enthusiasts off the Barrier Reef. The story focuses in part on the desperate search of the father to rescue his son, Nemo. A similar story can be told of origin of life researchers who are striving to rescue the notion that the life-giving molecules known as amino acids can be spontaneously produced from inorganic sources.
Back in 1952, Stanley Miller, with the assistance of Harold Urey, conducted an experiment which tickled our curiosity with their ground breaking apparatus that was designed to recreate an early-earth environment. A chemical mixture (H20, NH3, CH4 and H2) in a reducing atmosphere thought to be representative of that environment was heated and given electric sparks (“lightning”) and amino acids emerged! The fanfare over the significance of this discovery was quickly tempered, though, because it only produced a small number of the 20 amino acids essential for life.

Later adjustments to this mixture were made and amongst the different iterations, they produced over 40 different amino acids. Unfortunately, only ten of those were ones needed in living systems. In addition to the inability to account for these missing amino acids, the significance of this research was further confounded with the realization that the early earth atmosphere contained oxygen. In the presence of oxygen any amino acids that might have been produced would quickly be decomposed.
To solve the problem of the missing amino acids, origin of life researchers along with astrobiologists have turned their attention toward space. Extraterrestrial material discovered on earth has been found to possess some amino acids, but it can’t be certain whether their presence can be attributed to contamination from the earth environment. So, space probes are being sent to asteroids to sample their contents in space. Even if scientists are able to identify the presence of amino acids in the samples from these asteroids (which would be very interesting), an extraterrestrial source would not have been able to produce the quantity of amino acids needed to overcome the dilution factors present on earth and meet the demands for the origin of life.

To solve the oxygen problem, it has been suggested the one place on earth with a reducing atmosphere sufficient for amino acid production is volcanic vents on the ocean floor. In early 2019, researchers at NASA’s Jet Propulsion Laboratory created a model of a volcanic vent in the laboratory which yielded the amino acid alanine (also produced in the Miller-Urey experiment). While this proof-of-concept experiment is of some interest, it fails to have much geochemical relevancy. The lab simulation used very controlled conditions (temperature and pH). The chemical environment was created using pure reagents with a specified mixture which would not be expected to be found at volcanic vents.
In the natural environment, an untold number of chemical impurities would interfere with these reactions, and not result in the same outcome. An additional problem with this scenario is that the amino acids produced in this environment would not persist very long. At the temperatures which may be found at volcanic vents (600° F) the half-life of amino acids is only a few minutes.
A further complication with deep-sea production of amino acids is the lack of potential for these amino acids to form proteins. Because of the nature of the peptide bonding reaction, proteins cannot be synthesized in a watery environment. The amino acids must be relocated to a place where the concentrating and drying of amino acids can occur which is a long way off from the bottom of the ocean. It is most probable that any amino acids produced from volcanic vents would suffer the fate of dilution in the vast ocean.
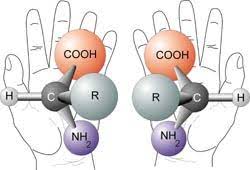
One more hurdle for moving from amino acids to living cells is that of amino acid selection. There are over 500 naturally occurring amino acids. Each amino acid comes in two forms which are mirror images of each other (i.e. chirality or stereo isomers). In laboratory production of amino acids, both types of amino acids are produced in equal quantities known as a racemic mixture. In living systems, however, only 20 amino acids are specified in the genetic code, and only the left-handed versions of these amino acids seem to be useful.
It is often discussed how exceedingly unlikely it would be to produce biologically relevant proteins (requiring hundreds of amino acids each) by just randomly assembling the 20 particular amino acids. When one realizes that, a) there are actually over a thousand amino acids from which to select, and b) there are no chemical reaction scenarios that can bias this selection, the protein problem becomes intractable.
In the seven decades since Miller and Urey’s experiment, the gap in our understanding of how amino acids originated and became incorporated into living systems has simply gotten wider. What should not go unnoticed from all the work of origin of life researchers is that it requires a lot of concentrated mental effort to design, plan and create controlled conditions which result in amino acids, and then also separate out the ones important for biomolecules. Beyond just raw materials and environmental conditions, it seems that an essential element for the existence of amino acids is intelligent agency.

I recognize that some may be put off by such an inference – that it is somehow inappropriate to appeal to transcendent forces when explaining our universe – that it is a case of a “god of the gaps” argument, and we ought to simply take a position of agnosticism. Some would further object and say that while we do know amino acids can be formed, we just have not figured out yet how it happened in early earth’s history – scientists will eventually figure it out.
The first objection is largely a philosophical commitment, and not one necessitated by science itself. Science as a discipline is charged with determining the function and formation of the world around us. The emphasis has been to focus on the operation of natural laws, but in understanding natural causation, we should also be able to detect supernatural causation. If natural laws do not suffice to explain a phenomenon, but agent causation can be demonstrated to produce the observed effect, agent causation is not excluded by default as an explanation.

The second objection seems to me an appeal to wishful thinking. As people gained insight into elemental properties they realized, for instance, that lead cannot be transformed into gold if we simply rely on natural laws and conditions (a human-designed particle accelerator, though, might do the trick). When it comes to understanding how inorganic molecules are transformed into amino acids and proteins, we are not hampered by a lack of information. We have sufficient insight into the chemical nature of amino acid formation and terrestrial environmental conditions to exclude spontaneous formation of amino acids to explain the origin of life. Appealing to agent causation for an explanation here is not a “god of the gaps argument”, but appealing to the work of science is – the “god” in this case is science itself.
Nemo’s father was eventually reunited with his long-lost son with lots of effort and many lessons learned along the way. For origin of life researchers, however, the prospects are dim for finding a means of producing amino acids (and proteins) through the operation of natural laws alone. Through their efforts, though, enough lessons should be learned by now to direct our attention to design as a better explanation than chance and necessity. That realization should not stop the work of origin of life researchers – it should only change the way they approach the problem. An openness to design principles along with the understanding of natural laws would hopefully lead to even greater insights.
Suggested Resources:
Origins of Life by Fazale Rana and Hugh Ross (NavPress, 2004)
James Tour, professor of chemistry and nano-engineering at Rice University discusses claims regarding origin of life research https://www.youtube.com/watch?v=r4sP1E1Jd_Y&t=941s