“Water is our very best friend, but at the same time it is our very worst enemy.” This is the conundrum I present my students with when learning about the importance of water for life on our planet. One of water’s fine qualities is its action as a solvent – the best one on the planet. For instance, water can dissolve important minerals like nitrogen, phosphorus and potassium which makes them accessible to living things in an ecosystem. This same characteristic, however, prevents molecules from coalescing – the very thing needed for cells to take shape. How do living systems overcome this destructive nature of water? The answer is found in a uniquely structured molecule called a phospholipid.
A phospholipid is a molecule composed of two fatty acid chains and a phosphate group linked together by a three-carbon molecule called glycerol. The two ends of this molecule have opposite characteristics: the end with the phosphate group is hydrophilic (water-loving) while the end with the two fatty acid chains is hydrophobic (water-fearing). Within a cell membrane, which is largely composed of phospholipids, these molecules arrange themselves into a phospholipid bilayer such that the fatty acid chains move away from water and orient themselves toward the center of the bilayer while the phosphate ends are oriented toward the inner and outer surface of the membrane. The effect of this arrangement is that the cell membrane formed with this bilayer can interact in a watery environment (both inside and outside the cell), and take advantage of things like water’s ability to transport nutrients. At the same time, the fatty acid chains within the bilayer prevent water from moving in amongst these molecules so a cell can maintain its structure. Brilliant!
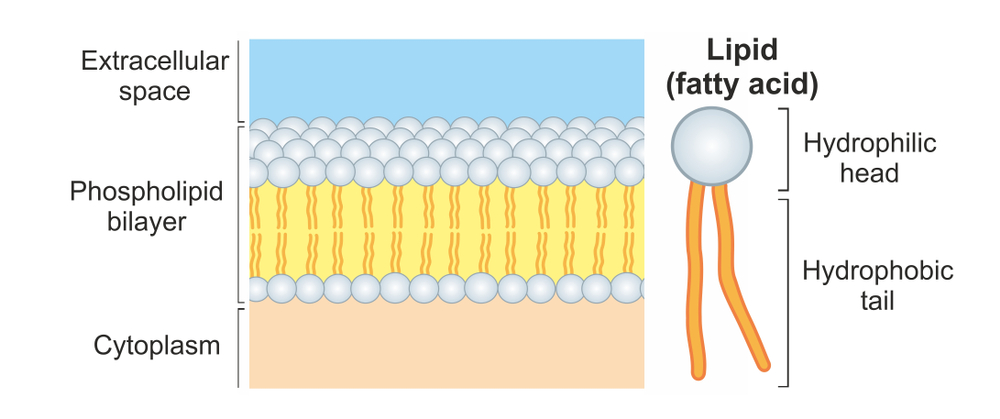
Where do cells come by these remarkable little water-resistant molecules? They make them! The fatty acid chains are first constructed by enzymes in the cytoplasm using two-carbon molecules derived from the metabolism of sugar. Then, five different membrane bound enzymes within the cellular organelle known as the smooth endoplasmic reticulum catalyze a series of reactions which attach the fatty acid chains and a phosphate molecule to the glycerol molecule. Bottom line: this is a complex process requiring many specialized proteins. But if cells are required to make phospholipids, where did the phospholipids come from to make the cell membranes of the first cells?
In the website Exploring Life’s Origins, (http://exploringorigins.org/fattyacids.html) the authors provide us with a highly speculative account about how the first protocells could have had membranes formed from simple fatty acids. They suggest that fatty acids could have formed in hydrothermal vents, and that membranes formed from these fatty acids could have developed on a certain type of clay particle some distance from the hydrothermal vents. However, what is depicted in this website regarding the formation of the first cell membranes are nothing but phantoms – things that we perceive, but have no physical reality. From the presentation on this website, our perception is garnered through imaginative story telling and computer animations, and we are left with a sense that cell membranes could have emerged on their own through natural processes. That perception, though, is not supported by any real evidence.
First of all, nobody has actually observed the formation of fatty acids in a hydrothermal vent. Further, the formation of highly organized lipid structures (e.g. liposomes) that could house cellular processes would require high concentrations of lipids under strictly controlled pH levels. There is no known mechanism which would accomplish this in an early earth environment. While they can be created in a laboratory setting, there is no evidence that liposomes formed from fatty acids and contained RNA ever existed in nature. Fatty acids will only persist for a few months, so any liposome that might have formed could not have lasted long enough to evolve into anything more than a pile of decomposing hydrocarbons.
More importantly, there is no account for the eventual formation of phospholipids. There are thousands of ways fatty acids and phosphates could be attached to a glycerol molecule, but only one will work for cell membranes. Scientists have been able to develop a carefully planned protocol for the inorganic production of phospholipids which involves the use of pure reagents, and a process of alternating the wetting and drying of materials under controlled temperature and pH conditions – none of which could have reasonably occurred in an early earth environment. What this strongly suggests is that the production of those first phospholipids was no accident, but required intelligent agency.
For Further Reading
If you would like to learn more about the challenges which face the formation of the first cell membranes, you might be interested in reading:
Creating Life in the Lab by Fazale Rana (Baker Books, 2011).
1 thought on “Phantoms and Phospholipids”