Evolutionary biologist, Theodosius Dobzhansky, is often quoted from his essay in 1973 in which he wrote, “Nothing in biology makes sense except in the light of evolution”. Since that time, it has become clear there are several aspects of life on earth of which evolution fails to make any sense. These aspects are all exemplified in the wings of insects. Two of these were examined in the previous articles in this series: first, the lack of any transitional forms in the fossil record which lead to the appearance of insects with wings, and second, the inability to account for anatomical novelty – especially in the case of insect wings which are comprised of multiple interdependent parts, each of which is an anatomical novelty themselves. A third aspect to be examined in this article is the origin of phenotypic complexity.
Darwin’s version of evolutionary theory depended heavily upon the notion of variability – the thing upon which natural selection is to act. He was at a loss, though, to explain the source of variation. A promising lead to this void in understanding was Gregor Mendel’s work on inheritance, and the presence of some factor within cells called genes that would bring about these traits. Since that time, science has provided tremendous insights into the causes for different traits (phenotypes) and how they might be varied by random mutations to the sequence of DNA bases which direct the outcome. But ongoing research has brought to light a picture of gene expression which is considerably more complex than anyone ever imagined – what is referred to as phenotypic complexity. As Marion, etal., describe it:
Most phenotypic components are comprised of multiple traits collectively operating as an integrated unit…Examples of complex phenotypes include behavioral syndromes, biomechanical systems, chemical defenses, and metabolic pathways. Complex phenotypes might characterize individual cells, organ systems, organisms, social groups, or ecosystems.[1]
Relating to insect flight, a genetic study published in 2011 attempted to determine the number of genes which were involved in controlling the wing shape of the fruit fly (Drosophila melanogaster). By studying the effects of all the laboratory induced mutations of fruit flies, the researchers noted that a high percentage of the fruit fly genome influenced wing architecture. In particular, they identified 94 candidate genes which influence the shape of the wing.[2] Each of these genes was identified because a mutation to that gene caused a deformity in the wing. The researchers were careful to specify that this number does not include the many other genes which control the size of the wing. Some of these mutations have more direct effects than others. Some genes for instance control things like how well cells stick to each other and communicate with each other – genes that influence many more traits than just the wing shape.
Not only is evolution unable to account for phenotypic complexity, it likewise is unable to account for new genetic information which would be necessary to explain the origin of insect wings. Mutations which are known to occur in DNA are only able to act on existing information and modify existing phenotypes. To produce new information, one speculation has been that accidental gene duplication which can occur during replication could provide the genetic resources for new genes. It is suggested the extra gene copy could then be modified for other functions without losing the information from the original gene. That is much easier said than done.
Mutations to a hypothetical gene copy are random, and have no eye to future needs of the organism – it doesn’t “know” what it should make of this extra piece of DNA. But even if the gene could be modified into something useful, additional genetic information beyond the gene itself would need to be present to make it functional. Every gene has a promoter region to which signaling proteins will bind and result in the transcription and translation of the protein for which the gene codes. In other words, the gene needs to have a way for it to be recognized by the cell. Otherwise, it would be like a mis-shelved book in the library – it might have useful information, but it can’t be found to be used. In addition to that, if a new gene is formed it needs to have a way to be recruited into the overall regulatory program of the cell or organism: when and where will this gene be expressed. A very useful gene could actually be harmful if it was expressed in the wrong place or the wrong time.
Even more problematic is the consequence of mutations to genes which are involved in phenotypically complex expression. A recent paper described the application of a mathematical model which used information theory to help quantify the complexity of phenotypes. The model operates under the assumption that the higher incidence of strongly harmful mutations in more complex phenotypes holds uniformly across all genotypes with such a phenotype.[3] In other words, the more complex the phenotype, the more likely you are to develop a harmful mutation. This creates a significant barrier to developing complex biological systems as the risk of a trait being damaged grows with its complexity.
Evolutionary theory comes up empty handed when it attempts to explain the origin of insect flight. Biologists posit very weak scenarios for their evolutionary origin which have no evidence to support them. The theory cannot account for the source of new information that is essential for the existence of anatomical novelties. Nor is the theory able to explain the origin of highly complex structures which require the simultaneous development and integration of complex subcomponents. We do have experience, however, which tells us how such things may come about.
The new information required for anatomical novelty is not just any information. It is a type of information known as specified complexity. To be “specified” means that it conforms to a known pattern (e.g. a sequence of base pairs can be translated into a particular sequence of amino acids which fold into a functional protein). The “complexity” of this information involves its probability – the more complex it is, the lower its existence can be accounted for by chance. As William Dembski explains, “the specified complexity exhibited in living forms convincingly demonstrates that blind natural forces could not by themselves have produced those forms but that their emergence also required the contribution of a designing intelligence.”[4] In every instance of information that entails specified complexity, the source of that information is intelligent agency.
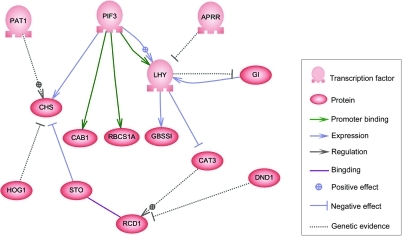
This same cause is the known source for complex regulatory networks. In our day we use computer programs to operate integrated systems such as robotic manufacturing, automobile engines and railway networks. Nobody would presume these computer programs were produced by randomly generated computer code. What we find within cells is a very complex regulatory network that specifies which genes are expressed in each type of cell and when they should be expressed in order to accurately generate complex phenotypes like the shape of an insect wing. It seems most reasonable that such networks were established, not by random mutations, but by intelligent agency.
This same cause again is the known source for the development and construction of highly complex machinery. The function of an automobile engine and transmission requires a purposeful arrangement of complex parts which is best attributed to intelligent design. As Michael Behe writes, “the effects of a mind can be recognized by the purposeful arrangement of parts, even when the possessor of the mind that arranged them is nowhere to be seen.”[5] It is a much more reasonable approach to presume that intelligent agency is behind human technologies than to presume they came about by any kind of random process. It is no less reasonable to presume that some intelligent agency is responsible for the purposeful arrangement of complex parts assembled to accomplish insect flight.

[1] Zachary H. Marion, James A. Fordyce, and Benjamin M. Fitzpatrick, “Extending the Concept of Diversity Partitioning to Characterize Phenotypic Complexity,” The American Naturalist 186, no. 3 (September 2015): 348-361. https://doi-org.ezproxy.biola.edu/10.1086/682369 (accessed 05/01/20).
[2] Carreira, Valeria P et al. “Genetic basis of wing morphogenesis in Drosophila: sexual dimorphism and non-allometric effects of shape variation.” BMC developmental biology vol. 11, no. 32 (02/06/2011), doi:10.1186/1471-213X-11-32 (accessed 04/29/20).
[3] Andreas Wagner, “Information theory and the phenotypic complexity of evolutionary adaptations and innovations,” bioRxiv (08/22/2016). https://doi.org/10.1101/070854 (accessed 4/27/2020). This assumption for the model was indicated in this preprint version of the journal article. This assumption was left out from the article when it was published as: Andreas Wagner, “Information theory, evolutionary innovations and evolvability” Phil. Trans. R. Soc. B37220160416 http://doi.org/10.1098/rstb.2016.0416 (accessed 4/27/20).
[4] William Dembski, The Design Revolution: Answering the Toughest Questions About Intelligent Design (Downers Grove, IL: IVP, 2004), 147.
[5] Michael Behe, Darwin Devolves: The New Science About DNA That Challenges Evolution (New York: Harper Collins, 2019), 264.
1 thought on “On the Wings of Insects (pt 3)”